
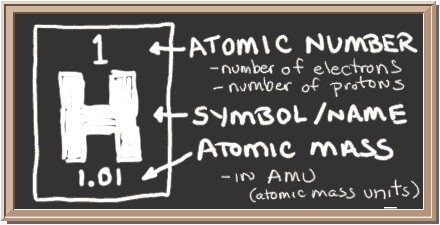
Check out the blackboard. That box on the left has all of the information you need to know about one element. It tells you the mass of an average atom (relative atomic mass), how many pieces are inside, and where it should be placed on the periodic table.
So what does the image tell you? - Hydrogen has the symbol H.
- With the atomic number one, there is one proton in the nucleus and one electron orbiting the nucleus of a normal (electrically neutral) hydrogen atom.
- The relative atomic mass minus the atomic number tells you there are no neutrons in the nucleus of an average atom. Isotopes of atoms can have different numbers of neutrons.
Shell number one can only hold 2 electrons, shell two can hold 8, and for the first eighteen elements shell three can holds eight electrons. (2-8-8) As you learn about elements with more than eighteen electrons you will find that shell three can hold more than eight. Once a shell is full, the next electron that is added must move to the next outer shell.
So... for the element of HYDROGEN, you already know that the atomic number tells you the number of electrons. That means there is only one electron in a normal hydrogen atom. iLooking at the picture, you can see there is only one electron in shell one.
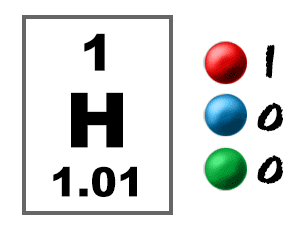
So what does the image tell you? - Hydrogen has the symbol H.
- With the atomic number one, there is one proton in the nucleus and one electron orbiting the nucleus of a normal (electrically neutral) hydrogen atom.
- The relative atomic mass minus the atomic number tells you there are no neutrons in the nucleus of an average atom. Isotopes of atoms can have different numbers of neutrons.
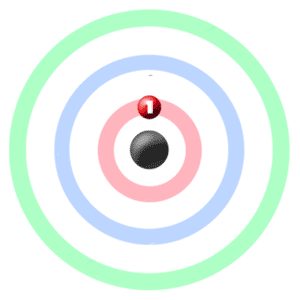
Electrons in the Shells
Take a look at the picture below. Each of those colored balls is an electron. In an atom, the electrons spin around a central area called the nucleus. Electrons like to be in separate shells/orbitals.Shell number one can only hold 2 electrons, shell two can hold 8, and for the first eighteen elements shell three can holds eight electrons. (2-8-8) As you learn about elements with more than eighteen electrons you will find that shell three can hold more than eight. Once a shell is full, the next electron that is added must move to the next outer shell.
So... for the element of HYDROGEN, you already know that the atomic number tells you the number of electrons. That means there is only one electron in a normal hydrogen atom. iLooking at the picture, you can see there is only one electron in shell one.


Compounds of Hydrogen (Basic Examples)
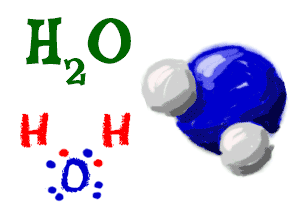
Water
Even though you find complex names on other molecules, everyone calls H2O "water". Water is made up of two hydrogen (H) atoms and one oxygen (O) atom. The formula for water is H2O. The hydrogen atoms have filled orbitals with two electrons and the oxygen atom has a filled second orbital with eight electrons.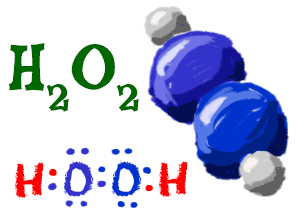
Hydrogen Peroxide
Two hydrogen atoms can also bond with two oxygen (O) atoms (peroxide), making the formula H2O2. There is only one more oxygen atom than water (H2O) but it is a totally different compound. You can see that each of the oxygen atoms has eight electrons and the two hydrogens have two electrons each. See how the electrons are shared?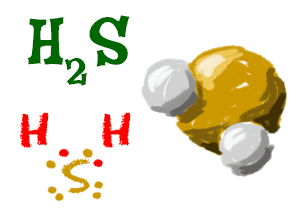
Hydrogen Sulfide
Two hydrogen atoms can also bond with one sulfur (S) atom and make the compound H2S (also known as hydrogen sulfide). The same way that oxygen (O) is happy when it has two extra electrons, sulfur likes to have two extras as well. See how the sulfur atom gets to share each of the electrons from the hydrogen atoms?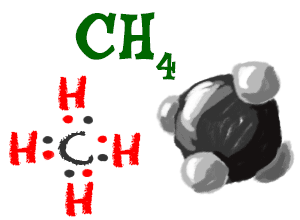
Methane
Hydrogen can also bond with carbon (C) in a huge number of molecules called alkanes. The simplest example is methane (CH4) with one carbon atom and four hydrogen atoms. The carbon atom fills its second shell with eight electrons and each of the four hydrogen atoms has two electrons.
► HYDROGEN BASICS
► HYDROGEN QUIZ & VIDEOS
► PREVIOUS ELEMENT
► NEXT ELEMENT
► RETURN TO TOP OF PAGE
► Or search the sites...
► HYDROGEN QUIZ & VIDEOS
► PREVIOUS ELEMENT
► NEXT ELEMENT
► RETURN TO TOP OF PAGE
► Or search the sites...


