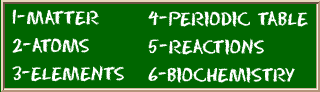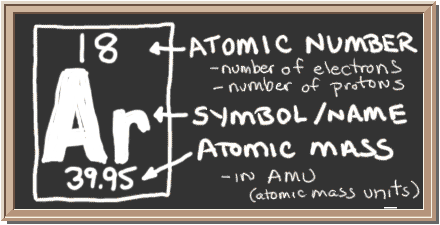
Check out the blackboard. That box on the left has all of the information you need to know about one element. It tells you the mass of one atom, how many pieces are inside, and where it should be placed on the periodic table.
In the next section we're going to cover electron orbitals or electron shells. This may be a new topic to some of you.
Electrons In The Shells
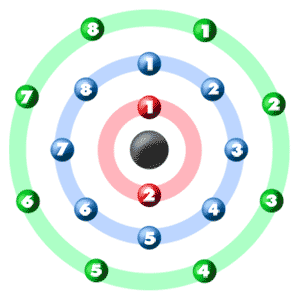
Take a look at the picture below. Each of those colored balls is an electron. In an atom, the electrons spin around the center, also called the nucleus. The electrons like to be in separate shells/orbitals. Shell number one can only hold 2 electrons, shell two can hold 8, and for the first eighteen elements shell three can hold a maximum of eight electrons. As you learn about elements with more than eighteen electrons you will find that shell three can hold more than eight. Once one shell is full, the next electron that is added has to move to the next shell.So... for the element of ARGON, you already know that the atomic number tells you the number of electrons. That means there are 18 electrons in an argon atom. Looking at the picture, you can see there are two electrons in shell one, eight in shell two, and eight in shell three.
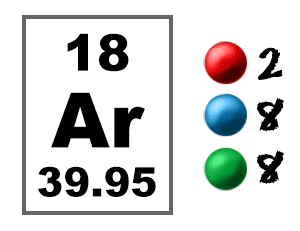
Examples of Compounds with Argon
ArgonArgon is the third of the noble gases or inert gases. It is very non-reactive. So much so, that it forms compounds with virtually no other elements. Just like neon (Ne) and helium (He), argon (Ar) usually floats around all by itself.It is non-reactive because the shells are full. Argon has three electron shells. The third shell is filled with eight electrons. That is why it does not easily combine with other elements. |
|
|