Halogens on the Right
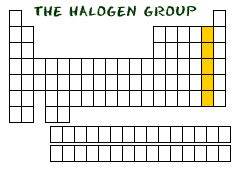 In the second column from the right side of the periodic table, you will find Group Seventeen (Group XVII). This column is the home of the halogen family of elements. Who is in this family? The elements included are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At).
In the second column from the right side of the periodic table, you will find Group Seventeen (Group XVII). This column is the home of the halogen family of elements. Who is in this family? The elements included are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At).
What Makes Them Similar?
When you look at our descriptions of the elements fluorine and chlorine, you will see that they both have seven electrons in their outer shell. That seven-electron trait applies to all of the halogens. They are all just one electron shy of having full shells. Because they are so close to being happy, they have the trait of combining with many different elements. They are very reactive. You will often find them bonding with metals and elements from Group One of the periodic table. The elements in the column on the left each have one electron that they like to donate. We've just told you how reactive the halogens are. Not all halogens react with the same intensity or enthusiasm. Fluorine is the most reactive and combines with most elements from around the periodic table. Reactivity decreases as you move down the column. As you learn more about the table, you will find this pattern true for other families. As the atomic number increases, the atoms get bigger. Their chemical properties change just a little bit when compared to the element right above them on the table.
We've just told you how reactive the halogens are. Not all halogens react with the same intensity or enthusiasm. Fluorine is the most reactive and combines with most elements from around the periodic table. Reactivity decreases as you move down the column. As you learn more about the table, you will find this pattern true for other families. As the atomic number increases, the atoms get bigger. Their chemical properties change just a little bit when compared to the element right above them on the table.
What is a Halide?
The elements we are talking about in this section are called halogens. When a halogen combines with another element, the resulting compound is called a halide. One of the best examples of a halide is sodium chloride (NaCl). Don't think that the halogens always make ionic compounds and salts. Some halides of the world are a part of molecules with covalent bonds.Related Video...
Earth Science: Salt of the Earth (NASA/GSFC Video)