Building Proteins from Amino Acid Chains
We already showed you some information about
amino acids.
Proteins are made of amino acids. Even though a protein can be very complex, it is basically a long chain of amino acid subunits all twisted around like a knot.
Primary Structure
As proteins are being built, they begin as a straight chain of amino acids. This chain structure is called the primary structure. Sometimes chains can
bond to each other with two
sulfur (S) atoms. Those bonds would be called a
disulfide bridge.
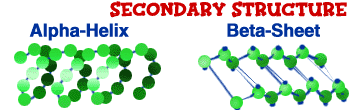
Secondary Structure
After the primary structure comes the secondary structure. The original chain begins to twist. It's as if you take a piece of string and twist one end. It slowly begins to curl up. In the amino acid chain, each of the amino acids interacts with the others and it twists like a corkscrew (
alpha helix) or it takes the shape of a folded sheet (
beta sheet). We talked about amino acids that are
hydrophobic and
hydrophilic. Those desires to stay away or be close to water (H
2O) play a part in the twisting.
Tertiary Makes Step Three
Let's move on to the tertiary structure of proteins. By now you're probably getting the idea that proteins do a lot of folding and twisting. The third step in the creation of a protein is the tertiary structur. The amino acid chains begin to fold even more and bond using more bridges (the disulfide bridges).
Quaternary Is Fourth and Final
We can finally cover the quaternary structure of proteins. Quaternary means four. This is the fourth phase in the creation of a protein. In the quaternary structure, several amino acid chains fromthe tertiary structures fold together in a blob. You heard us right. "Blob" is the term we use on this site. They wind, entwined, in and out of each other. Some of the most famous protein blobs are
hemoglobin in human red blood cells and the
photosystems in plant chloroplasts.
Related Video...
3D Proteins: The Big Picture (US-NSF Video)