
Sweet, Sweet Carbs
 Carbohydrate is a fancy way of saying "sugar." Scientists came up with the name because the molecule have many carbon (C) atoms bonded to hydroxide (OH-) groups. Carbohydrates can be very small or very large molecules, but they are still considered sugars. Plants can create long chains of these molecules for food storage or structural reasons.
Carbohydrate is a fancy way of saying "sugar." Scientists came up with the name because the molecule have many carbon (C) atoms bonded to hydroxide (OH-) groups. Carbohydrates can be very small or very large molecules, but they are still considered sugars. Plants can create long chains of these molecules for food storage or structural reasons.
What's It Used For?
A carbohydrate is called an organic compound, because it is made up of a long chain of carbon atoms. Sugars provide living things with energy and act as substances used for structure. When sugars are broken down in the mitochondria, they can power cell machinery to create the energy-rich compound called ATP (adenosine triphosphate). Some examples of structural uses might be the shell of a crab (chitin) or the stem of a plant (cellulose). We'll talk about them in a little bit.Saccharides
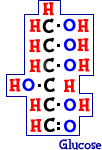 Scientists also use the word saccharide to describe sugars. If there is only one sugar molecule, it is called a monosaccharide. If there are two, it is a disaccharide. If there are three, it is a trisaccharide. You get the idea.
Scientists also use the word saccharide to describe sugars. If there is only one sugar molecule, it is called a monosaccharide. If there are two, it is a disaccharide. If there are three, it is a trisaccharide. You get the idea.
Simple Sugars
What about the simplest of sugars? A sugar called glucose is the most important monosaccharide on Earth. Glucose (C6H12O6) is created by photosynthesis and used in cellular respiration. When you think of table sugar, like the kind in candy, it is actually a disaccharide. The sugar on your dinner table is made of glucose and another monosaccharide called fructose (C6H12O6). These sugars have the same numbers of atoms, but they are different structures called isomers.Polysaccharides
When several carbohydrates combine, it is called a polysaccharide ("poly" means many). Hundreds of sugars can be combined in a branched chain. These chains are also known as starches. You can find starches in foods such as pasta and potatoes. They are very good sources of energy for your body.Sugars for Structure and Support
 An important structural polysaccharide is cellulose. Cellulose is found in plants. It is one of those carbohydrates used to support or protect an organism. Cellulose is in wood and the cell walls of plants. You know that shirt you're wearing? If it is made of cotton, that's cellulose, too! There can be thousands of glucose subunits in one large molecule of cellulose. If we were like some herbivores or insects, such as termites, we could eat cellulose for food. Those animals don't actually digest the polysaccharides. They have small microorganisms in their bellies that break down the molecules and release smaller sugars.
An important structural polysaccharide is cellulose. Cellulose is found in plants. It is one of those carbohydrates used to support or protect an organism. Cellulose is in wood and the cell walls of plants. You know that shirt you're wearing? If it is made of cotton, that's cellulose, too! There can be thousands of glucose subunits in one large molecule of cellulose. If we were like some herbivores or insects, such as termites, we could eat cellulose for food. Those animals don't actually digest the polysaccharides. They have small microorganisms in their bellies that break down the molecules and release smaller sugars.
Polysaccharides are also used in the shells (chitin) of crustaceans, such as crabs and lobsters. Chitin is similar in some ways to the structure of cellulose, but has a far different use. The shells are solid, protective structures that need to be molted (left behind) when the crustacean begins to grow. It is very inflexible. On the other hand, it is very resistant to damage. While a plant may burn, it takes very high temperatures to hurt the shell of a crab. If you know the way crabs are cooked, you know that the crab meat cooks on the inside of the shells when it is boiled. There is no damage to the shells at the temperature of boiling water (H2O at 100oC).
► NEXT PAGE ON BIOCHEMISTRY
► NEXT STOP ON SITE TOUR
► BIOCHEMISTRY QUIZ
► RETURN TO TOP OF PAGE
► Or search the sites...
► NEXT STOP ON SITE TOUR
► BIOCHEMISTRY QUIZ
► RETURN TO TOP OF PAGE
► Or search the sites...
Related Video...
Chalk Talk: Liquid Crystals (US-NSF Video)



