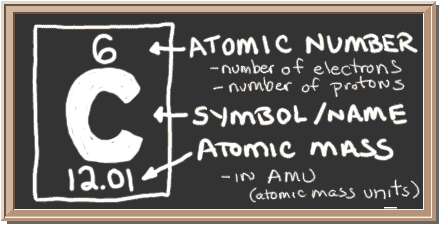
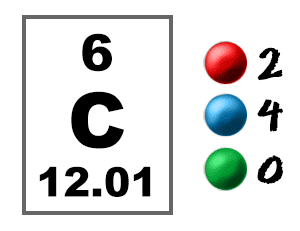
Check out the blackboard. That box on the left has all of the information you need to know about one element. It tells you the mass of one atom, how many pieces are inside, and where it should be placed on the periodic table.
In the next section we're going to cover electron orbitals or electron shells. This may be a new topic to some of you.
Electrons In The Shells
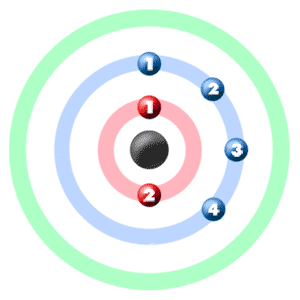
Take a look at the picture below. Each of those colored balls is an electron. In an atom, the electrons spin around the center, also called the nucleus. The electrons like to be in separate shells/orbitals. Shell number one can only hold 2 electrons, shell two can hold 8, and for the first eighteen elements shell three can hold a maximum of eight electrons. As you learn about elements with more than eighteen electrons you will find that shell three can hold more than eight. Once one shell is full, the next electron that is added has to move to the next shell.So... for the element of CARBON, you already know that the atomic number tells you the number of electrons. That means there are 6 electrons in a carbon atom. Looking at the picture, you can see there are two electrons in shell one and four electrons in shell two.
Examples of Compounds with Carbon
Carbon DioxideThis is a carbon dioxide molecule. When you breathe out, you usually breathe out carbon dioxide. With the formula CO2 that means there are two oxygen (O) atoms and one carbon (C) atom. If you look closely at the dot structure, you'll see that they share four electrons each. If a bond shares two electrons that means it is a single bond. If a bond is made up of four electrons it is a double bond. That means that the carbon atom has two double bonds, one with each oxygen atom. |
|
|
|
|
||
Cyanogen ChlorideHere's something new! We have three different elements here, carbon (C), nitrogen (N), and chlorine (Cl). That's not special, but the way they combine is! Look at the carbon and the nitrogen, they are sharing six electrons!When two atoms share two electrons, that's a single bond. If they share four it's a double bond. Well these two are sharing six, that's a triple bond. It's extremely strong and powerful. It would take a lot of work to separate the C and the N! One more thing! Because the bond between carbon and nitrogen is so strong, scientists call them "cyanogen" instead of carbon-nitrogen. Scientists know that cyanogen is always CN. |
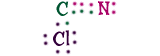 |
|
|
|
||
Beryllium CarbideTwo beryllium (Be) atoms are able to bond with one carbon (C) atom to create Be2C. The beryllium atoms let the carbon use their electrons so that the carbon is 'happy'. Each beryllium gives up both of its two extra electrons to the carbon. Take a look and see how all of the electrons are shared. |
|
|